What is Electronic Health Information?
What is Electronic Health Information?
It is important to clarify what we understand the term electronic health information (EHI) to represent within the broader context of electronic data and information about health.
EHI refers to an individual’s health data that are collected, maintained, and stored electronically by healthcare providers (HCPs) and healthcare organizations (HCOs) (AHIMA, 2021; ONC, 2022). This can also include certain data held by public health authorities (PHAs), such as immunization records, surveillance data, and vital records. For a more technical definition of EHI see Office of the National Coordinator’s (ONC) Understanding Electronic Health Information (EHI).
It is important to clarify what we understand the term electronic health information (EHI) to represent within the broader context of electronic data and information about health.
EHI refers to an individual’s health data that are collected, maintained, and stored electronically by healthcare providers (HCPs) and healthcare organizations (HCOs) (AHIMA, 2021; ONC, 2022). This can also include certain data held by public health authorities (PHAs), such as immunization records, surveillance data, and vital records. For a more technical definition of EHI see Office of the National Coordinator’s (ONC) Understanding Electronic Health Information (EHI).
Electronic Health Records
Electronic Health Records
Electronic health records (EHRs) are, at their simplest, digital versions of patients’ paper charts and can be real-time, patient-centered records. They provide immediate access to various types of clinical and patient information, “whenever and wherever it is needed” (ONC, 2022). EHRs collate information about a patient’s health (ASTP/ONC, 2018), including patient history, diagnoses, medications, immunization records, allergies, radiology images, and laboratory test results.
Electronic Laboratory Reporting
Electronic Laboratory Reporting
Electronic Laboratory Reporting (ELR) is the transmission of digital laboratory reports via a standards-based message format from laboratories to HCPs and PHAs. ELR automates the reporting process by translating information into an electronic message that can be automatically sent and processed. ELR is a critical tool in ensuring rapid and accurate reporting to PHAs for case investigation and identification of outbreaks (CDC, 2024). ELR is often the first notification a PHA receives on a reportable condition. The information contains minimal demographic and laboratory results, requiring additional provider follow-up for information that is often contained in EHRs.
Electronic Case Reporting
Electronic Case Reporting
Electronic case reporting (eCR) is the automated process of exchanging disease report submission information for reportable conditions from HCPs to PHAs. Reportable conditions are diseases or other conditions that HCPs, laboratories, or other entities are legally required to report to PHAs (CSTE, 2007). These reports help PHAs track disease trends, implement control measures, and prevent outbreaks. The specific conditions that are reportable vary by jurisdiction (state, territory, or local) and are determined by laws and regulations. The eCR system uses EHRs to facilitate the secure transfer of data, streamlining the process of disease tracking, case management, and contact tracing (CDC, 2024). eCR provides PHAs with comprehensive demographic and clinical information on reportable conditions, reducing manual and paper–based methods of information exchange.
The diagram below depicts the relationship between EHI, EHR, ELR, eCR and public health.
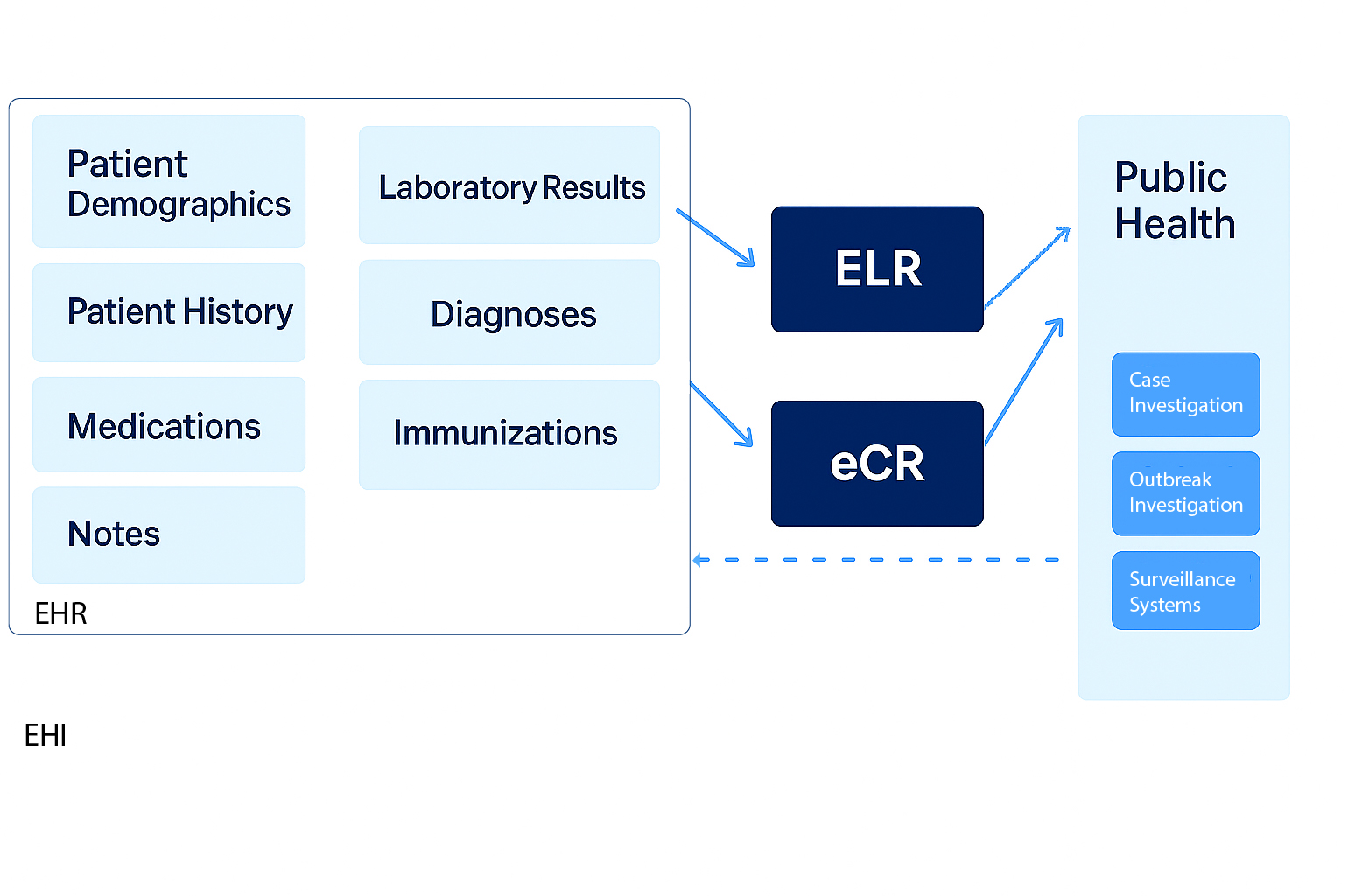
Alt text: Diagram showing how EHI relates to EHR data such as demographics, history, medications, notes, labs, diagnoses, and immunizations flow through ELR and eCR to public health for case investigation, outbreak investigation, and surveillance systems.
Toolkit Navigation
References
AHIMA (2021). EHI Task Force Report: Defining EHI and the Designated Record Set in an Electronic World. https://www.ahima.org/media/ztqh1h2q/final-ehi-task-force-report.pdf Accessed June 2, 2025.
Assistant Secretary for Technology Policy (ASTP)/Office of the National Coordinator for Health Information Technology (ONC) (2018). What are Electronic Health Records (EHRs)? https://www.healthit.gov/topic/health-it-and-health-information-exchange-basics/what-are-electronic-health-records-ehrs#:~:text=EHRs%20are%2C%20at%20their%20simplest,payer%20requirements%20and%20consumer%20expectations. Accessed June 2, 2025.
Centers for Disease Control and Prevention (CDC) (2024). Electronic Case Reporting (eCR): What is eCR? https://www.cdc.gov/electronic-lab-reporting/php/about/index.html#:~:text=What%20is%20ELR?,reports%20between%20public%20health%20agencies. Accessed June 2, 2025.
Centers for Disease Control and Prevention (CDC) (2024). Electronic Laboratory Reporting (ELR): What is ELR? https://www.cdc.gov/electronic-lab-reporting/php/about/index.html#:~:text=What%20is%20ELR?,reports%20between%20public%20health%20agencies. Accessed June 2, 2025.
Council of State and Territorial Epidemiologists (CSTE) (2008). Position Statement 08-EC-02: Criteria for Inclusion of Conditions on CSTE Nationally Notifiable Condition List and for Categorization as Immediately or Routinely Notifiable. https://cdn.ymaws.com/www.cste.org/resource/resmgr/PS/08-EC-02.pdf#:~:text=A%20reportable/notifiable%20condition%20is%20one%20for%20which,to%20local%20or%20state%20public%20health%20authorities. Accessed June 24, 2025.
Office of the National Coordinator for Health Information Technology (ONC) (2022). USCDI ONDEC (ONC New Data Element and Class) Submission System. https://www.healthit.gov/isp/ONDEC Accessed June 2, 2025.
Office of the National Coordinator for Health Information Technology (ONC) (2021). Understanding Electronic Health Information (EHI). www.healthit.gov/sites/default/files/page2/2021-12/Understanding_EHI.pdf Accessed Jun 25, 2025.








