Operationalizing EHI Access
Enhancing Public Health Practice using EHI
Electronic Health Information (EHI) contains essential clinical data that, when accessed appropriately, can significantly improve the timeliness, accuracy, and effectiveness of public health practice—including surveillance and response efforts.
By integrating structured and timely EHI data elements—such as laboratory results, diagnoses, treatments, vaccination status, and demographic details—public health professionals can enhance a wide range of activities.
Below are several common use cases demonstrating how EHI can be leveraged to strengthen public health practice.
Use Cases for Incorporating EHI
EHI can provide information critical for public health investigation of cases of reportable conditions and outbreaks, including but not limited to, history of present illness, past medical history, social and travel history, medications and vaccinations, laboratory and radiologic results, and clinical impressions and assessments.
Case Verification and Classification Use Case
Case verification and classification is used to confirm whether reported cases meet standardized public health case definitions, enabling consistent classification and counting across jurisdictions. Relevant EHI data elements could include laboratory results, diagnosis codes, clinical notes, patient demographics, and encounter details. Because EHI provides real-time access to lab and diagnostic data, the PHA avoids delays and reduces the need to re-contact providers for verification. It can also allow public health to gather important detailed information on risk factors for illness, which can be used to help public health professionals determine who is most at risk and how to protect them.
For example, a state public health agency receives an automated feed of potential hepatitis A cases from multiple healthcare organizations. To verify and classify these cases epidemiologists:
- Access EHI to review lab results for hepatitis A IgM antibodies, which are required for case confirmation under the case definition
- Cross-reference diagnosis codes and clinical notes to ensure symptoms (e.g., jaundice, elevated liver enzymes) align with the definition
- Check whether the patient was tested and whether results are pending or unavailable for cases missing lab confirmation,
- If the EHI confirms the case meets the established case definition, it is classified as a confirmed case
- If the data are incomplete or ambiguous, the case may be classified as probable, suspect, or flagged for follow-up
- With confirmed HAV cases, employment history may have been collected by the HCO and accessing this through the EHR could prompt immediate PH intervention for those who work in certain sectors (e.g., food service or childcare workers)
Case Investigation Use Case
Case investigation is used to identify, track, and manage individuals who may have been exposed to a communicable disease, and to understand the transmission dynamics of an outbreak. Relevant EHI data elements could include patient demographics, clinical encounters, symptoms and diagnoses, laboratory results, treatment history, vaccination status, and contact information.
For example, during a measles outbreak, public health investigators use EHI to:
- Identify confirmed cases based on lab results and diagnosis codes
- Determine the onset date and location of exposure
- Assess vaccination status and identify unvaccinated individuals
- Trace contacts based on shared healthcare encounters or household addresses
- Monitor outcomes and complications to guide clinical and public health response
Public Health Emergency Response Use Case
EHI can provide PHAs with immediate access to data contained in EHRs during mass exposure events (i.e., contaminated food, bioterrorism, etc.). This can allow the PHA to gain a better understanding of how an event or response activities are progressing, along with detailed clinical and sometimes exposure or risk information. Access to EHI during a public health emergency facilitates rapid contact tracing or prioritized testing of suspected cases. Surveillance work and case ascertainment during public health emergency response can be done in remote or off-site settings, which improves staff efficiencies and improves timely public health response where needed.
For example, a freight train derails near a suburban neighborhood, releasing hazardous chemicals into the air and water supply. Initial reports to the PHA include emergency calls and environmental monitoring data but lack detailed clinical information about affected individuals.
Access to EHI can assist the PHA and first responders by:
Supplementing limited reports: Public health professionals access EHI to review emergency department (ED) visits and EMS records from nearby hospitals. These records reveal symptoms consistent with chemical exposure—such as respiratory distress, dizziness, and skin irritation—that were not captured in initial reports to the PHA.
Identifying Additional Cases: By querying EHRs for patients presenting with relevant symptoms and ICD-10 codes, PHAs identify additional individuals who sought care but were not initially linked to the event.
Understanding Exposure and Outcomes: EHI provides timestamps, geographic data, and clinical details that help epidemiologists:
- Map exposure zones based on patient addresses and visit times
- Assess severity by reviewing lab results (e.g., elevated liver enzymes, abnormal blood counts)
- Track outcomes such as hospitalization, recovery, or long-term follow-up needs
Reducing Delays: Immediate access to EHI eliminates the need to re-contact healthcare organizations for verification, allowing PHAs to act quickly to issue public advisories, deploy mobile response units, and coordinate environmental remediation.
Syndromic Surveillance/Early Signal Detection Use Case
PHAs use EHI to obtain information about patterns of disease and risk within the community through early signal detection activities such as syndromic surveillance. Syndromic surveillance can be based on many types of data including ICD codes, defined syndromes, laboratory test results, and even risk factors. Increases in reports of specific data, such as chief complaints provided as part of ED visits, can provide PHAs with an early signal that there is a problem within a community.
For example, a heat-related illness spike occurs during an extended summer heatwave. In mid-July a PHA in a large metropolitan area begins monitoring ED data related to the heatwave. Syndromic surveillance systems flag a sudden increase in emergency department visits with chief complaints such as “dizziness,” “heat exhaustion,” “dehydration,” and “fainting.”
How EHI enhances early detection in this example:
- Provides real-time access to ED chief complaints and ICD-10 codes (e.g., T67.0 for heatstroke, R55 for syncope). Analysts notice a 3x increase in heat-related codes compared to the same period last year.
- Analyzing patient demographics and ZIP codes, PHAs identify that most cases are concentrated in low-income neighborhoods with limited access to air conditioning or cooling centers.
- Risk factor identification – revealing that many affected individuals are elderly, have chronic conditions (e.g., cardiovascular disease), are taking medications that impair thermoregulation, or are young men working in manual labor jobs.
- Tracking outcomes through clinical notes and lab results showing elevated body temperatures, abnormal electrolyte levels, and hospital admissions for severe dehydration and kidney stress.
Early detection allows the PHA to prioritize new cooling stations in neighborhoods that indicate an increase in heat related illness and lack access to air conditioning.
Outbreak Investigation and Exposure Follow Up Use Case
Public health professionals investigating outbreaks and following up on exposures can use EHI to supplement limited information contained in initial reports to better understand the nature of the outbreak or exposure and the risk to the public. Access to EHI can also lead to the identification of additional cases who might have been previously undetected. Immediate access to EHR data can provide PHAs with critical information about both exposures and clinical outcomes during large scale events.
For example: EHI can be used during an outbreak investigation of a surge in respiratory disease cases in a long-term care facility by supporting:
- Immediate access to clinical data: The PHA accesses EHRs from affiliated hospitals and clinics to review respiratory virus test results, including dates and outcomes; symptom onset and progression documented in clinical notes; comorbidities and risk factors; vaccination status, including vaccine type and administration dates; and hospitalization and intensive care unit (ICU) admissions, to monitor severity and outcomes.
- Contact Tracing & Prioritized Testing: Using EHI, the PHA identifies:
- Staff and residents with recent positive tests or symptoms related to the outbreak
- Individuals who had close contact with confirmed cases
- Unvaccinated or those with medical conditions that make them susceptible for adverse outcomes related to respiratory disease exposure and infection. T These individuals would be prioritized for testing and provided housing away from those who are ill.
Conceptual Workflow for Electronic Health Record Data Use in Surveillance
The diagram below shows a conceptual workflow of the process for using electronic health record (EHR) data in public health surveillance through a direct login to the HCO EHR. This process may differ if the PHA is accessing this data through a health information exchange (HIE).
The process may begin with a report of a notifiable condition occurring in several possible ways (see blue shapes). Regardless of how the PHA learns of the case the following steps are typically consistent.
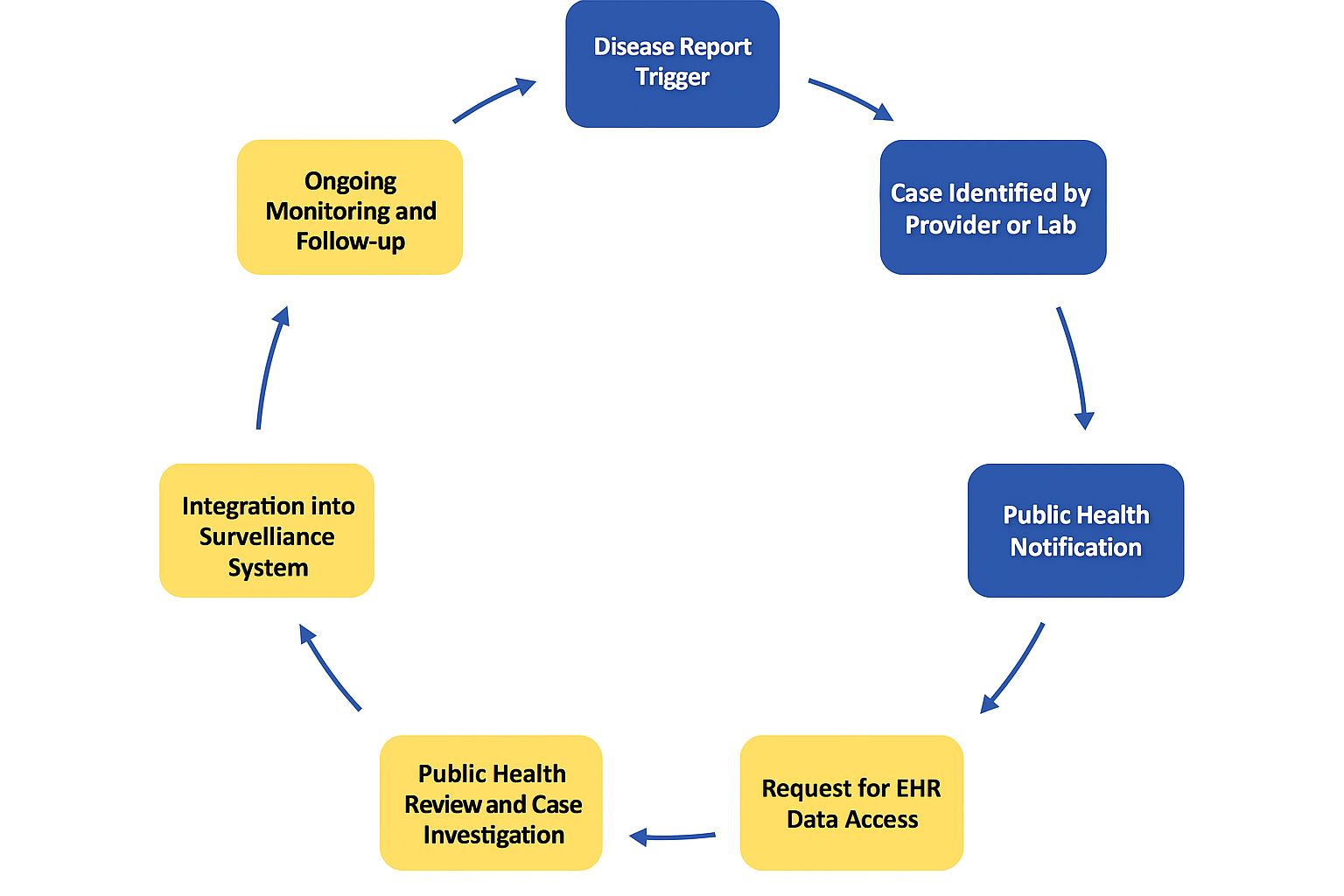
Alt text: A circular flowchart showing the disease reporting process with the following steps in order: Disease Report Trigger, Case Identified by Provider or Lab, Public Health Notification, Request for EHR Data Access, Public Health Review and Case Investigation, Integration into Surveillance System, and Ongoing Monitoring and Follow-up.








